The FDA is now under competing federal court orders regarding its approval of mifepristone.
Update April 13, 3:20 a.m. PST: In a divided opinion issued late Wednesday, the U.S. Court of Appeals for the Fifth Circuit ruled mifepristone will remain approved for use up to seven weeks of pregnancy but can no longer be sent in the mail while the case is being appealed. Clinicians can still prescribe the medication off label later in pregnancy, as many are already doing through 11 weeks gestation. The appeals court concluded that the plaintiffs had waited too long to challenge the 2000 approval in court but were timely on later modifications of the approval in 2016, 2019 and 2021 so allowed the Texas court ruling to stay in effect on these later modifications, including the 2019 approval of a generic version of mifepristone produced by GenBioPro.
Update April 10 at 2:49 p.m. PST: The Department of Justice filed a request for clarification with the Washington state court on its decision released last Friday, describing the court’s ruling as in “significant tension” with the Texas abortion ruling.
Massachusetts Governor Maura Healey announced the state has bought 15,000 doses of mifepristone—a two year supply—to “ride out” the legal challenge. Last week, Washington Governor Jay Inslee bought 30,000 doses of mifepristone—a three year supply for that state.
Over 250 pharmaceutical executives, including the CEO of Pfizer, have signed a letter objecting to the Texas ruling. Several major pharmaceuticals companies have not yet signed the letter, including Moderna, Johnson & Johnson and Eli Lily.

On Friday, April 7, federal judges in both Texas and Washington state issued contradictory rulings on the abortion pill mifepristone.
- Federal judge Matthew Kacsmaryk of the Northern District of Texas ruled that the FDA improperly approved mifepristone and said he would void the approval on April 14.
- One hour later, fellow federal judge Thomas O. Rice in the Eastern District of Washington issued an injunction blocking the FDA from “altering the status quo and rights as it relates to the availability of mifepristone” in the 17 plaintiff states and the District of Columbia.
The FDA is now under contradictory federal court orders regarding its approval of mifepristone—with one court saying the FDA approval of mifepristone is invalid, and another saying it must maintain its approval of mifepristone.
“FDA is under one order that says you can do nothing, and another that says in seven days I’m going to require you to vacate the approval of mifepristone,” Glenn Cohen of Harvard Law School told the Associated Press.
The Texas court also ruled that mailing abortion pills is a crime in violation of the 1873 Comstock Act, which prohibited sending obscene materials through the U.S. mail.
The Department of Justice immediately appealed Kacsmaryk’s ruling. Attorney General Merrick Garland said the DOJ “strongly disagrees with the decision. … [which] overturns the FDA’s expert judgment, rendered over two decades ago, that mifepristone is safe and effective. The department will continue to defend the FDA’s decision.”
Garland said the DOJ is reviewing Judge Rice’s decision.
“With two opposing rulings in place, the case will go quickly to the U.S. Supreme Court,” wrote Heather Cox Richardson in Letters from an American on Friday.
Kacsmaryk Decision Uses ‘Every Anti-Abortion Term Out There’
The Texas opinion is replete with anti-abortion misinformation, such as the false claim that abortion causes depression. “Women who receive abortions have a 154 [percent] higher risk of death from suicide than if they gave birth,” Kacsmaryk wrote—a claim disputed by the American Psychological Association. In reality, research from the Turnaway Study shows people denied abortions have worse physical and mental health than those who obtain them.
In the 67-page decision, Kacsmaryk referred to mifepristone as “chemical abortion” 93 times. Anti-abortion extremists have for years used the phrase “chemical abortion” to make abortion pills sound dangerous—when in fact they are very safe, even safer than Tylenol.
“Judge Kacsmaryk is just as ideologically motivated and a political actor as we feared. He uses every anti-abortion term and study out there,” said law professor and legal expert David S. Cohen on Twitter.
With no attempt to mask his bias, Kacsmaryk used inflammatory and medically inaccurate anti-abortion language.
- He called a fertilized egg, zygote and embryo an “unborn children,” saying the FDA “mandates … mifepristone to kill the unborn human.”
- He called a woman who has never given birth a “mother” because she is pregnant.
- He called clinicians “abortionists.”
“Matthew Kacsmaryk’s decision claiming to overrule the FDA’s approval of abortion drug mifepristone—23 years late—is an appalling and indefensible abuse of power that will go down in history as one of the judiciary’s most humiliating and lawless moments,” tweeted journalist Mark Joseph Stern.
Anti-Abortion Groups First Asked Kacsmaryk to Ban Mailing Abortion Pills in November
The ruling in the Texas case is a response to a federal lawsuit filed by abortion opponents in November 2022, challenging the FDA approval of the medication mifepristone in 2000—as well as subsequent updates to the approval in 2016, 2019 and most recently in December 2021, when the agency began allowing clinicians to mail abortion pills to patients.
The lawsuit alleged the FDA did not have proof of the medication’s safety—despite extensive evidence showing abortion pills are safe and effective.
Democratic AGs Filed Counter-Lawsuit to Protect and Expand Access to Abortion Pills
In the Washington state case, attorneys general of Arizona, Colorado, Connecticut, Delaware, District of Columbia, Illinois, Michigan, Nevada, New Mexico, Oregon, Rhode Island, Vermont, Hawaii, Maine, Maryland, Minnesota, Pennsylvania and Washington sued the FDA in February. The suit asked the federal court to declare that mifepristone is safe and effective and that the FDA’s approval of mifepristone is lawful and valid. They also asked the court to remove several current restrictions on the medication, including the requirement that clinicians and pharmacies must be certified to prescribe mifepristone. Plaintiffs argued that the latter restrictions were not based on science and that they burdened access to mifepristone.
On the first request, Rice entered a preliminary injunction enjoining the FDA from removing mifepristone from the market, but he denied the second request for a preliminary injunction directing the FDA to remove the existing restrictions on mifepristone.
Texas Judge Doesn’t Have Power to Ban Abortion Pills Nationwide, Say Legal Experts
According to David Cohen and other legal experts, a federal judge cannot remove an FDA-approved medication from the market. Federal law provides a process for removing an unsafe drug from the market and grants the FDA discretion to make a final decision as to the safety of a medication—not federal judges.
“The judge doesn’t have the authority to withdraw approval for a drug. Congress has a very specific procedure in the Food, Drug and Cosmetic Act for withdrawing drug approvals,” Cohen told Ms. last month. “He may order the FDA to follow the withdrawal process, but that withdrawal process leaves the final decision in the FDA’s hands. So if there’s any wiggle room in the judge’s order, they could interpret the ruling as a requirement to start this process. And then they start that process. That can be a lengthy process with them as the final decision maker.”
Reviving Comstock
In a separate part of the Texas decision, Kacsmaryk suggested that mailing abortion pills is a crime in violation of the 1873 Comstock Act, which banned sending obscene literature, contraceptives, abortifacients or any sexual information through U.S. mails.
The DOJ last month issued its own opinion, ahead of the Friday ruling, that mailing abortion pills does not violate Comstock.
Legal experts say the Texas ruling has limited applicability. “Judge Kacsmaryk’s ruling that Comstock bans the mailing of abortion pills binds no one other than the FDA. NO ONE ELSE in the country is required to follow that ruling,” tweeted David Cohen.
“Women’s historians have been warning for decades that we never took the Comstock Act off the books. And now here we are,” tweeted historian Lara Freidenfelds.
In 2020, medication abortion accounted for 54 percent of all pregnancy terminations in the U.S. After a federal court lifted the FDA requirement that clinicians distribute the medication in person in July of 2020, telemedicine abortion services surged across the United States, becoming available in over 20 states, expanding further after the FDA permanently lifted the in-person distribution requirement in December of 2021.
After the Supreme Court reversed Roe v. Wade last June, abortion pills and telemedicine abortion became a critical way women were able to end pregnancies in states restricting abortion access. The Kacsmaryk decision puts this all at risk.
Fighting Back Against Kacsmaryk’s Ruling
Reproductive rights advocates question the enforceability of the lone Texas judge’s ruling.
Sen. Ron Wyden (D-Ore.) is urging the FDA and the Biden administration to ignore Kacsmaryk’s decision. “President Biden can and must ignore this ruling and keep mifepristone on the market and accessible for every woman in America,” tweeted Wyden.
In a senate floor speech in February, Wyden explained: “Congress long ago empowered the FDA, a body made up of scientists and clinicians, to approve or disapprove the use of new drugs—not states and certainly not activist judges. The FDA approved mifepristone 23 years ago. For those looking to challenge that approval—well, it’s too late. The statute of limitations allows challenges to FDA procedures for only six years.”
“Now let him enforce it,” journalist Dahlia Lithwick dared Kacsmaryk.
Reproductive Health Providers Continue to Offer Abortion Pills
Despite the Texas ruling, many telemedicine clinicians are still determined to provide abortion pills by mail, including mifepristone, to patients.
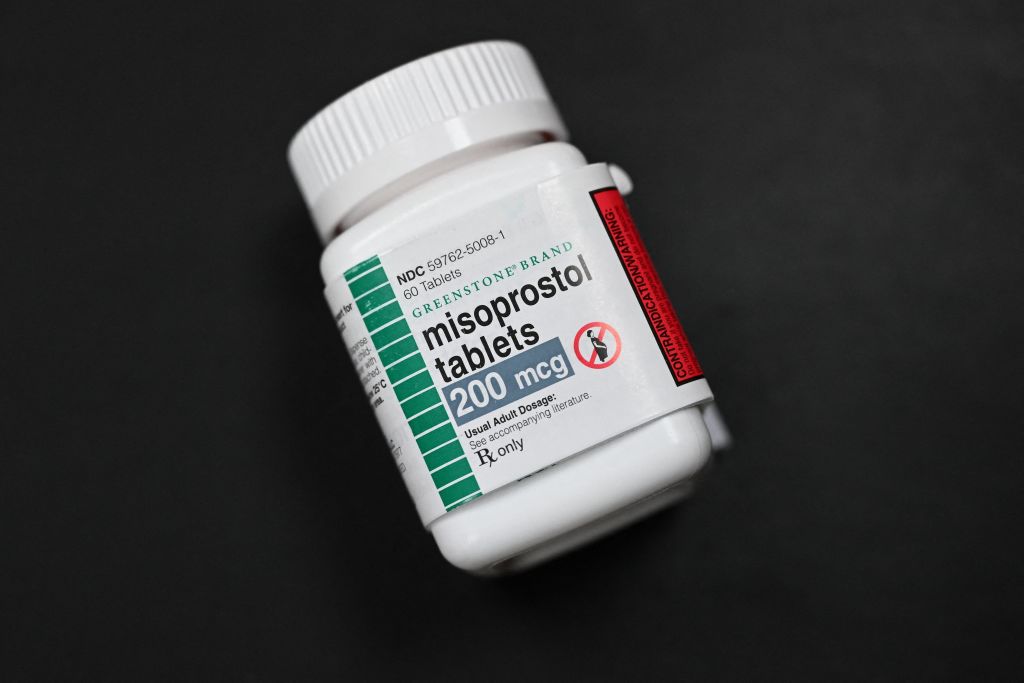
“Let me be clear: Mifepristone is extremely safe, effective, and still legally available through Hey Jane,” said Kiki Freedman, co-founder and CEO of Hey Jane, a telemedicine abortion provider. “Our focus remains on our patients and delivering the best possible evidence-based, compassionate care and therefore we will continue providing our current medication abortion protocol of mifepristone and misoprostol.”
Reproductive health clinicians are also ready, if need be, to offer a safe and effective alternative to end pregnancy through three months: a higher dosage of misoprostol taken alone. Misoprostol is a widely available ulcer medication that can induce a miscarriage by causing contractions of the uterus to expel a pregnancy. Many studies from around the world have found that self-managed abortion with misoprostol alone is 93 to 99 percent effective and very safe.
Laws Are Not Stopping People From Accessing Abortion Pills
Despite abortion restrictions and bans, people are obtaining abortion pills online. The organization Plan C has a comprehensive guide to finding abortion pills on their website, which is continually updated and has all the latest information on where to find abortion pills from anywhere in the U.S.
The Austria-based telemedicine abortion provider Aid Access serves people in all 50 states. Patients of all ages can obtain abortion pills, including advance provision, for a sliding scale fee of $110.
Meanwhile, legal experts are fighting back to maintain medication abortion access through the medical system in the United States.
“If our opponents think we will stand by as they steamroll medical ethics and jeopardize patients’ health and safety with their ideological extremism, they should think again,” said Deirdre Schifeling, national political director of the ACLU. “We will use every tool at our disposal until everyone can access the safe and essential reproductive healthcare they need.”
Up next:
U.S. democracy is at a dangerous inflection point—from the demise of abortion rights, to a lack of pay equity and parental leave, to skyrocketing maternal mortality, and attacks on trans health. Left unchecked, these crises will lead to wider gaps in political participation and representation. For 50 years, Ms. has been forging feminist journalism—reporting, rebelling and truth-telling from the front-lines, championing the Equal Rights Amendment, and centering the stories of those most impacted. With all that’s at stake for equality, we are redoubling our commitment for the next 50 years. In turn, we need your help, Support Ms. today with a donation—any amount that is meaningful to you. For as little as $5 each month, you’ll receive the print magazine along with our e-newsletters, action alerts, and invitations to Ms. Studios events and podcasts. We are grateful for your loyalty and ferocity.